- Updated on September 24, 2019
![]() By Dr. Artour Rakhimov, Alternative Health Educator and Author
By Dr. Artour Rakhimov, Alternative Health Educator and Author
 Glutamine is the most abundant and most required amino acid in the human organism (hence its popularity in bodybuilding). It is also the amino acid that is most needed for tissue repair. However, “since the supply of glutamic acid from the circulating blood is insufficient for the formation of additional amounts of glutamine, the dicarboxylic acid has to be synthesized in the brain” (Berl et al., 1962). This last substance is a CO2 derivative. Its production depends on CO2 levels in the brain and our breathing patterns.
Glutamine is the most abundant and most required amino acid in the human organism (hence its popularity in bodybuilding). It is also the amino acid that is most needed for tissue repair. However, “since the supply of glutamic acid from the circulating blood is insufficient for the formation of additional amounts of glutamine, the dicarboxylic acid has to be synthesized in the brain” (Berl et al., 1962). This last substance is a CO2 derivative. Its production depends on CO2 levels in the brain and our breathing patterns.
Many ordinary people – and all people with chronic diseases – have low body-oxygen levels due to deep automatic breathing (chronic hyperventilation) 24/7. Why is this so?
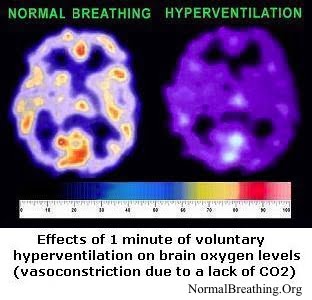 Overbreathing, as shown in dozens of studies, leads to reduced oxygen transport to brain cells and cells of other vital organs in the human body. This, in turn, leads to an anaerobic acidic environment and the production of free radicals causing oxidative damage to the brain and other tissues and organs. As a result, people experience increased glutamine demands for cell repair and insufficient glutamine synthesis due to low levels of CO2 in body cells. Why do CO2 levels matter?
Overbreathing, as shown in dozens of studies, leads to reduced oxygen transport to brain cells and cells of other vital organs in the human body. This, in turn, leads to an anaerobic acidic environment and the production of free radicals causing oxidative damage to the brain and other tissues and organs. As a result, people experience increased glutamine demands for cell repair and insufficient glutamine synthesis due to low levels of CO2 in body cells. Why do CO2 levels matter?
 A review of numerous research studies devoted to this subject was given by Waelsch and colleagues (1964) in an article entitled “Quantitative aspects of CO2 fixation in the mammalian brain in vivo.” They found that aspartic and glutamic amino acids and glutamine were substances that were chemically synthesized in mammalian brains. But CO2 is used to synthesize glutamine (Rossi et al., 1962; Waelsch et al., 1964; Pincus, 1968; Pincus et al., 1969; Cheng, 1971; Konitzer et al., 1977; Cheng et al., 1978; Tachiki & Baxter, 1980; Lockwood & Finn, 1982; Martin et al., 1992; Oz et al., 2004). You can read abstracts of some of these studies at the bottom of this page.
A review of numerous research studies devoted to this subject was given by Waelsch and colleagues (1964) in an article entitled “Quantitative aspects of CO2 fixation in the mammalian brain in vivo.” They found that aspartic and glutamic amino acids and glutamine were substances that were chemically synthesized in mammalian brains. But CO2 is used to synthesize glutamine (Rossi et al., 1962; Waelsch et al., 1964; Pincus, 1968; Pincus et al., 1969; Cheng, 1971; Konitzer et al., 1977; Cheng et al., 1978; Tachiki & Baxter, 1980; Lockwood & Finn, 1982; Martin et al., 1992; Oz et al., 2004). You can read abstracts of some of these studies at the bottom of this page.
All these studies suggest that the gas we exhale plays a crucial role in glutamine synthesis. Various other studies have found that cell hypocapnia (low CO2) causes the production of free radicals, oxidative stress, chronic inflammation, poor repair of injuries, and many other harmful effects (see CO2 links below).
 Thus, managing CO2 levels can be a way for the human organism to rebuild nerve tissue in the brain. The rate of CO2-derived glutamine production is proportional to the CO2 concentration in the brain. It can be concluded that low CO2 in the brain not only makes the brain unreasonably excited (often causing anxiety, insomnia, fears, panic attacks, aggression, hostility, violence, or other strong emotions), but also has adverse effects on the cellular repair.
Thus, managing CO2 levels can be a way for the human organism to rebuild nerve tissue in the brain. The rate of CO2-derived glutamine production is proportional to the CO2 concentration in the brain. It can be concluded that low CO2 in the brain not only makes the brain unreasonably excited (often causing anxiety, insomnia, fears, panic attacks, aggression, hostility, violence, or other strong emotions), but also has adverse effects on the cellular repair.
As a result, slow and light diaphragmatic nasal breathing leads to higher oxygen levels in the brain and body cells, favoring effective glutamine synthesis and normalizing numerous other chemical reactions.
If you want to read physiological evidence (abstracts of studies) for the CO2 role in glutamine synthesis and fixation in the brain, click here. The links and titles (details) of these studies are provided below.
References: Glutamine synthesis and CO2 fixation
Cheng SC. CO2 fixation in the nervous tissue. Int Rev Neurobiol. 1971;14:125-57.
Cheng SC, Naruse H, Brunner EA. Effects of sodium thiopental on the tricarboxylic acid cycle metabolism in mouse brain: CO2 fixation and metabolic compartmentation J Neurochem. 1978 Jun;30(6):1591-3.
Konitzer K, Voigt S, Hetey L. Carbon dioxide fixation in the brain: its relation to glucose synthesis. Acta Biol Med Ger. 1977; 36(2):147-56.
Lockwood AH, Finn RD. 11C-carbon dioxide fixation and equilibration in rat brain: effects on acid-base measurements.Neurology. 1982 Apr;32(4):451-4.
Martin G, Michoudet C, Vincent N, Baverel G. Release and fixation of CO2 by guinea-pig kidney tubules metabolizing aspartate. Biochem J. 1992 Jun 15; 284 (Pt 3): 697-703.
Oz G, Berkich DA, Henry PG, Xu Y, LaNoue K, Hutson SM, Gruetter R. Neuroglial metabolism in the awake rat brain: CO2 fixation increases with brain activity. J Neurosci. 2004 Dec 15;24(50):11273-9.
Pincus JH. The effect of atmospheric carbon dioxide on carbon dioxide fixation in rat brain. Neurology. 1968 Mar;18(3):293.
Pincus JH Carbon dioxide fixation in rat brain: Relationship to cerebral excitabilityExperimental Neurology, Volume 24, Issue 3, July 1969, Pages 339-347.
Rossi, CA, Berl, S., Clark, DD, Purpura, DP, and Waelsch, H. Rate of CO2 fixation in brain and liver. Life Sci. 1962 Oct;1:533-9.
Tachiki KH, Baxter CF. Role of carbon dioxide fixation, blood aspartate and glutamate in the adaptation of amphibian brain tissues to a hyperosmotic internal environment. Neurochem Res. 1980 Sep;5(9):993-1010.in blood but apparently not from CO2 fixation in the brain.
Waelsch H, Berl S, Rossi CA, Clarke DD, Purpura DP, Quantitative aspects of CO2 fixation in mammalian brain in vivo, J. Neurochem. Oct 1964, 11: 717-728.

